细胞核孔复合体是一种极其复杂的分子机器,其内部通道并非简单空洞,而由大量动态蛋白构成。研究人员通过构建与天然核孔复合体尺寸相同的合成孔道进行实验,发现当加入核孔蛋白(nucleoporins)和运输因子时,系统会形成一个中央“堵塞”结构,并表现出与酵母细胞天然核孔复合体相似的行为。这表明无序蛋白质结构在分子运输机制中发挥关键作用。
科学界仍在争论核孔通道的具体结构。部分理论认为内部呈凝胶状结构,另一些则认为类似刷状排列。最新建模研究提出混合模型:通道外围可能呈刷状结构,而中心区域则具有类似凝聚体或凝胶的性质。由于核孔结构不断变化,不同实验可能捕捉到不同状态,因此研究人员认为真实结构可能介于多种模型之间。
技术进步正在推动更精确的观察。2025年研究团队利用高分辨率三维成像技术Minflux追踪分子在人体细胞核中的运动,发现运输分子主要沿通道边缘移动。这一结果与中央区域可能存在“堵塞”结构的假设一致。核孔复合体对细胞生命过程至关重要,参与蛋白质合成与基因调控,但其结构弱点也使其成为多种疾病的关键靶点,包括癌症、病毒感染和神经发育疾病。
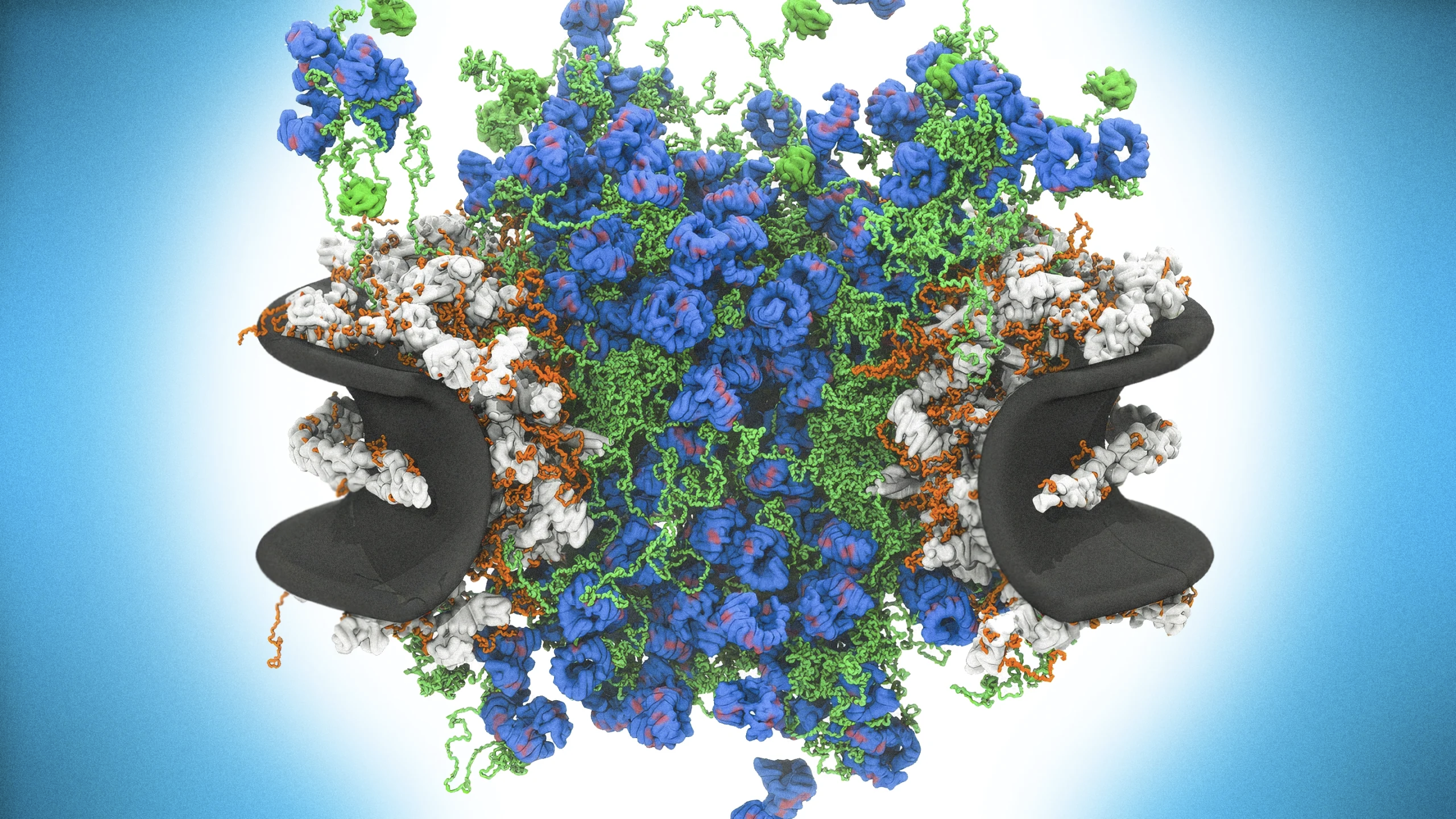
The nuclear pore complex is an extremely complex molecular machine whose inner channel is not an empty opening but a dense environment of dynamic proteins. Researchers built synthetic pores the same size as natural nuclear pore complexes and found that when nucleoporins and transport factors were added, the system formed a central “plug” structure and behaved similarly to nuclear pores in yeast cells. The results suggest that disordered protein structures play a crucial role in regulating molecular transport through the pore.
Scientists are still debating the exact structure of the transport channel. Some models propose that the interior behaves like a gel, while others describe a brush-like arrangement of proteins. Recent modeling studies suggest a hybrid structure in which the outer regions resemble a brush and the central region behaves more like a condensate or gel. Because the pore constantly changes configuration, different experiments may capture different states, indicating that the real structure may lie between multiple theoretical models.
Advances in imaging technology are enabling more detailed observations. In 2025 researchers used a high-resolution three-dimensional imaging method called Minflux to track molecules moving through nuclear pores in intact human cell nuclei and observed transport occurring mainly near the channel’s edges. This finding supports the possibility that a central plug may block the middle region. The nuclear pore complex is essential for cellular processes such as protein production and gene regulation, but its structural vulnerabilities also make it a target in diseases including cancer, viral infections, and neurodevelopmental disorders.